GemPharmatech Mouse Models Support Discovery in Cell Publication: Revealing a Novel Treg-Mediated Mechanism of Immunotherapy Resistance
Last December, GemPharmatech’s models were used in a publication accepted by the prestigious journal Cell.
The study led by Professors Ling Lu of Xuzhou Medical University and Professor Jian Gu of the First Affiliated Hospital of Nanjing Medical University titled, "Tumor-produced ammonia is metabolized by regulatory T cells to further impede anti-tumor immunity," reveals a fundamental new mechanism of tumor immune evasion. It demonstrates that tumor-produced ammonia is metabolically utilized by regulatory T cells (Tregs), enhancing their immunosuppressive capacity and driving resistance to treatment. In cancer immunotherapy, understanding how metabolic byproducts in the tumor microenvironment (TME) influence immune cells is a central focus.
Read below to learn how GemPharmatech's mouse models were instrumental in this research.
1. Experimental Design: Multidimensional Validation of Ammonia Metabolism Pathways
1.1 Innovative Application of Spatial Multi-Omics Technologies
The research team collected tumor tissue samples from six patients with hepatocellular carcinoma (HCC) and prepared frozen sections for analysis. Spatial gene expression profiling was performed using the Visium CytAssist platform, while adjacent sections were analyzed by AFADESI-MSI.
UMAP-based dimensionality reduction and clustering analyses enabled the identification of distinct immune cell–enriched regions. Despite intratumoral heterogeneity, regulatory T cells (Tregs) exhibited a significantly broader spatial distribution within tumor tissues.
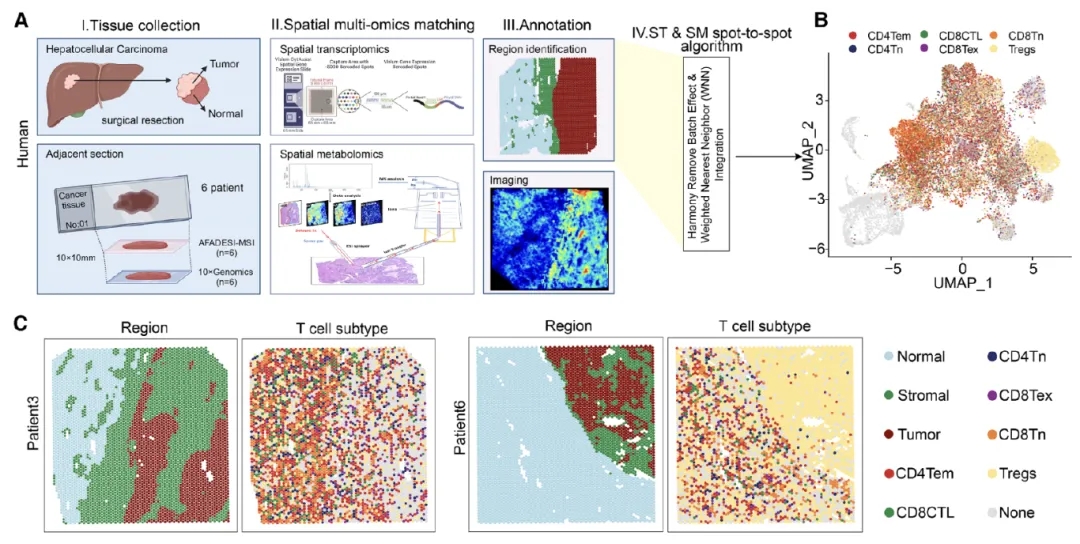
Figure 1. Tumor subregions are enriched in Treg cells, characterized by high glutamine catabolism and low urea cycle activity
1.2 Multilevel Validation Using Mouse Models
To validate the clinical observations, the researchers established an orthotopic hepatocellular carcinoma model in C57BL/6 mice using the Hep-53.4 cell line. Liver tumor tissues were collected at weeks 1, 2, and 3 following tumor cell injection for spatial metabolomic analysis, while adjacent sections were subjected to immunohistochemical staining for Tregs.
A series of genetically engineered mouse models were employed, including Asl fl/fl Foxp3YFP-Cre mice (Treg-specific Asl deletion), Pparr fl/fl Foxp3YFP-Cre mice (Treg-specific Pparg deletion), and PpargMut Foxp3YFP-Cre point-mutation mice, providing robust tools for mechanistic investigation.
In addition, multiple tumor models were examined, including subcutaneous tumor models (Hep-53.4, MC38, B16-F10, and Lewis cell lines), a tail-vein injection–induced HCC model (HTVI-HCC), and a metabolic dysfunction–associated steatohepatitis–HCC model (MASH-HCC). These complementary systems enabled comprehensive evaluation of ammonia metabolism across diverse tumor contexts.
2. Key Findings: Dual Ammonia Metabolism Pathways Enhance Treg Function
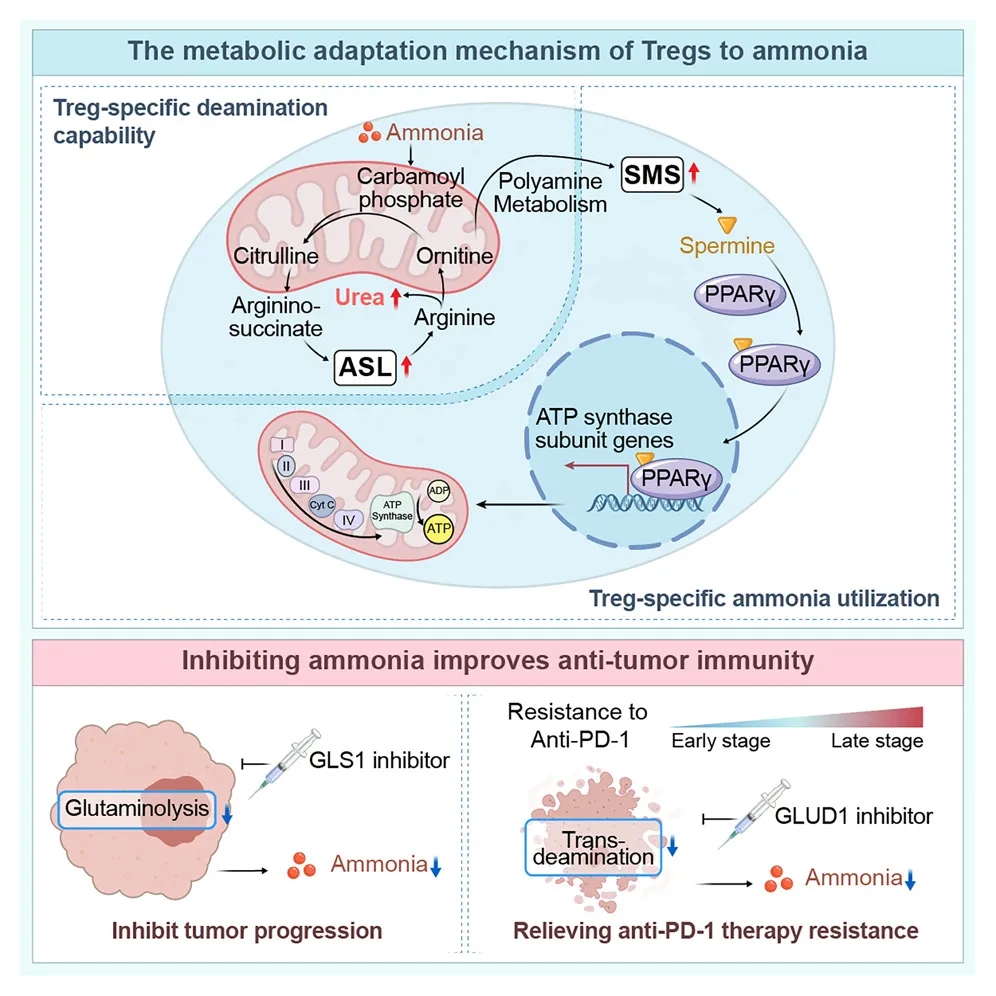
Figure 2. Adaptive ammonia metabolism mechanisms in regulatory T cells
2.1 Ammonia Induces Urea Cycle Activation in Tregs
Compared with CD8⁺ and CD4⁺ T cells, Tregs displayed markedly enhanced urea cycle activity under ammonia-rich conditions. 15N isotope tracing experiments demonstrated that Tregs upregulate argininosuccinate lyase (ASL) expression via SRC3-mediated STAT3 activation, thereby efficiently converting ammonia into urea cycle intermediates and alleviating ammonia-induced toxicity.
SRC3 was found to be selectively and highly expressed in Tregs. Structural modeling and surface plasmon resonance (SPR) analyses further revealed that ammonia enhances the binding affinity between SRC3 and STAT3, providing mechanistic insight into ammonia-driven signaling activation.
2.2 FOXP3-Driven Spermidine Synthase Expression Promotes Treg Function
The study further showed that, under transcriptional regulation by FOXP3, ammonia is converted into spermidine via spermidine synthase (SMS). X-ray crystallographic analyses demonstrated a direct interaction between spermidine and PPARγ, which broadly regulates the transcription of multiple mitochondrial respiratory chain complex components. This regulatory axis enhances oxidative phosphorylation in Tregs, thereby reinforcing their immunosuppressive activity.
2.3 Anti-PD-1 Therapy–Induced Ammonia Production Drives Treatment Resistance
Analysis of clinical samples revealed that HCC patients receiving anti-PD-1 therapy exhibited elevated intratumoral ammonia levels and increased Treg infiltration. Mechanistically, tumor cell death induced by anti-PD-1 treatment releases ammonia through transamination reactions, which in turn augments Treg function and contributes to immunotherapy resistance.
Consistent with these observations, animal studies demonstrated that combination treatment with the glutamate dehydrogenase (GLUD) inhibitor R162 and anti-PD-1 therapy significantly reduced intratumoral ammonia levels, limited Treg accumulation, enhanced CD8⁺ T-cell effector function, and effectively overcame resistance to PD-1 blockade.
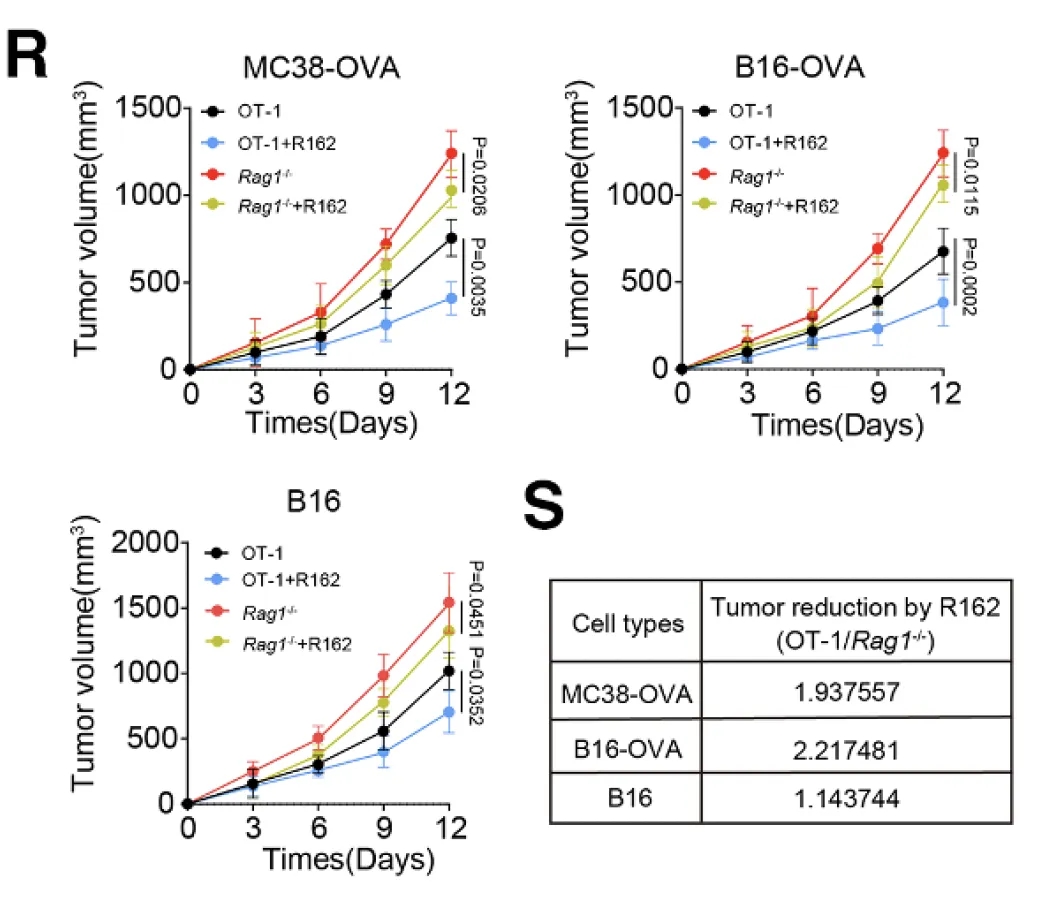
Figure 3. Pharmacodynamic evaluation of the GLUD1 inhibitor R162
This study not only elucidates the critical role of ammonia metabolism within the tumor microenvironment, but also identifies novel therapeutic targets and intervention strategies for overcoming resistance to cancer immunotherapy. The findings demonstrate that Tregs selectively adapt to ammonia stress through the SRC3/STAT3–ASL–mediated urea cycle pathway and the SMS–spermidine–PPARγ signaling axis, providing a strong theoretical foundation for the development of next-generation combination immunotherapies.
Targeting ammonia metabolism—particularly through the development of GLUD1 inhibitors—may represent a promising strategy to enhance the efficacy of PD-1 blockade and hold substantial translational potential.
GemPharmatech Mice Used for this Research Study:
Asl(fl/fl)Foxp3(YFP-Cre), Ppary(Mut)Foxp3(YFP-Cre) and Rag1-KO
As a leading provider of genetically engineered mouse models, GemPharmatech offers a comprehensive portfolio of KO/cKO mice, tool mice, humanized mice, immunodeficient mice, disease models and more.
Furthermore, we also offer model customization services to ensure a precise fit for specific research objectives. These resources provide robust support for pharmacodynamic evaluation and mechanistic studies in innovative drug development.
Is your research ready for the next in vivo phase? To facilitate this critical step, Now we offer a 25% discount on all custom model projects. This offer is valid through March 31, 2026. Contact us today to discuss your requirements and request a quote!