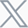
Comprehensive Preclinical Evaluation of STEAP1×CD3 T-cell Engager Using GemPharmatech’s Integrated Platform
STEAP1 is a highly attractive prostate cancer-specific antigen with restricted normal tissue expression, making it an ideal target for T-cell engager (TCE) immunotherapy. GemPharmatech established an integrated preclinical platform that combines in vitro functional assays with in vivo efficacy studies in humanized mouse models to comprehensively evaluate STEAP1×CD3 bispecific antibodies. The platform enables simultaneous assessment of antigen-dependent T-cell activation, cytotoxicity, tumor infiltration, antitumor efficacy, and cytokine profiles. Using the clinical-stage STEAP1×CD3 TCE Xaluritamig (AMG 509) as a benchmark, the system demonstrated robust coordinated evaluation capabilities, supporting translational development of next-generation T-cell engagers.
Download
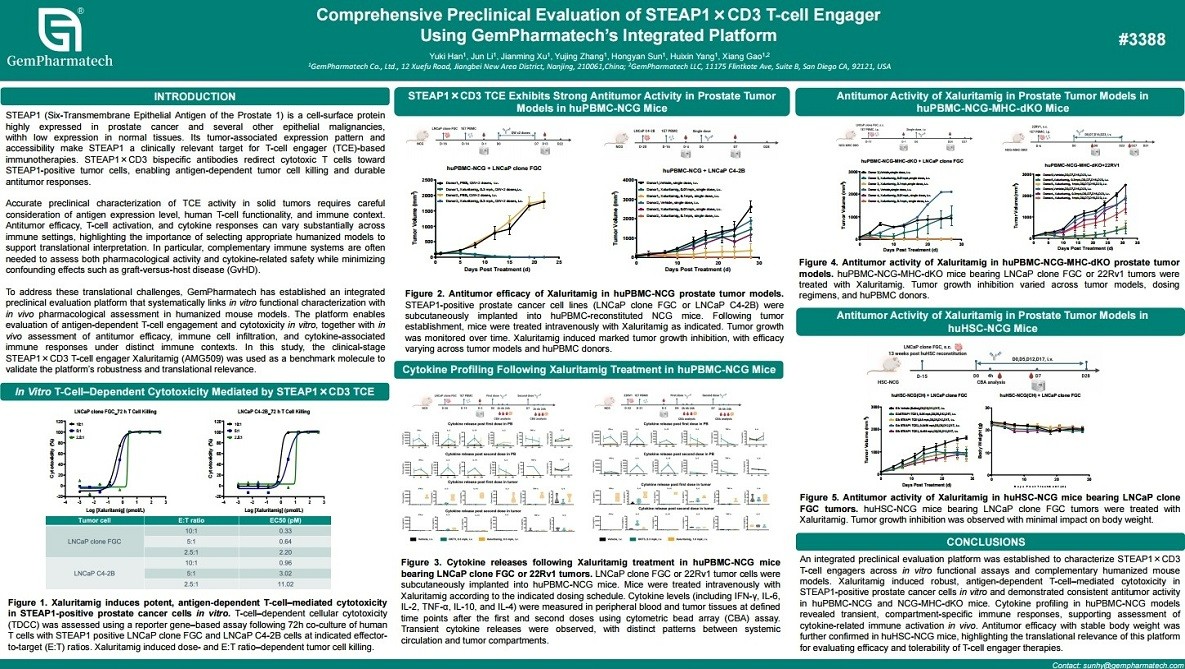
An Integrated Preclinical Platform for Comprehensive Evaluation of T-Cell Engagers
T-cell engagers (TCEs) are bispecific antibodies that simultaneously target CD3 on T cells and tumor-associated antigens, necessitating physiologically relevant models to accurately assess their mechanism of action. GemPharmatech has established an integrated preclinical platform that combines high-throughput in vitro assays for target binding, T-cell activation, and cytotoxicity with in vivo efficacy and safety studies in xenograft and syngeneic models. This platform utilizes NCG, NCG-MHC-dKO, and CD3-humanized mouse strains to enable comprehensive evaluation. Multiple TCE candidates demonstrated potent tumor growth inhibition, with efficacy closely correlated to target antigen expression and immune cell infiltration. This integrated platform provides a robust translational tool for advancing next-generation TCE in immuno-oncology.
Download
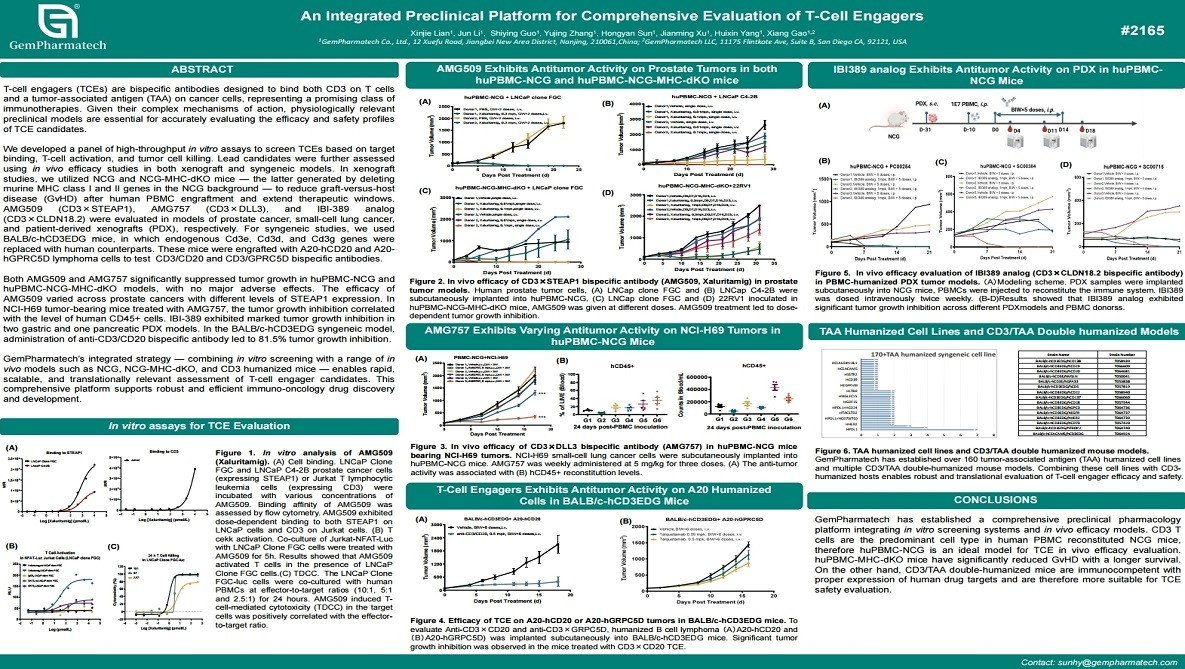
Novel T Cell Engager Targeting CD3 and CDH17 for the Treatment of Solid Tumors
Cadherin-17 (CDH17) is a highly promising target for T-cell engager (TCE) therapy due to its elevated expression in gastrointestinal tumors and restricted normal tissue distribution. However, conventional CD3-engaging TCEs are frequently limited by cytokine release syndrome (CRS). Using a fully humanized mouse platform, GemPharmatech identified cross-reactive CD3 and CDH17 binders and developed a novel CDH17×CD3 bispecific antibody. The lead candidate demonstrated potent, dose-dependent tumor cell killing comparable to a benchmark TCE, while inducing significantly lower levels of pro-inflammatory cytokines. This optimized TCE offers an improved therapeutic window and supports the translational development of safer bispecific antibodies for solid tumor treatment.
Download
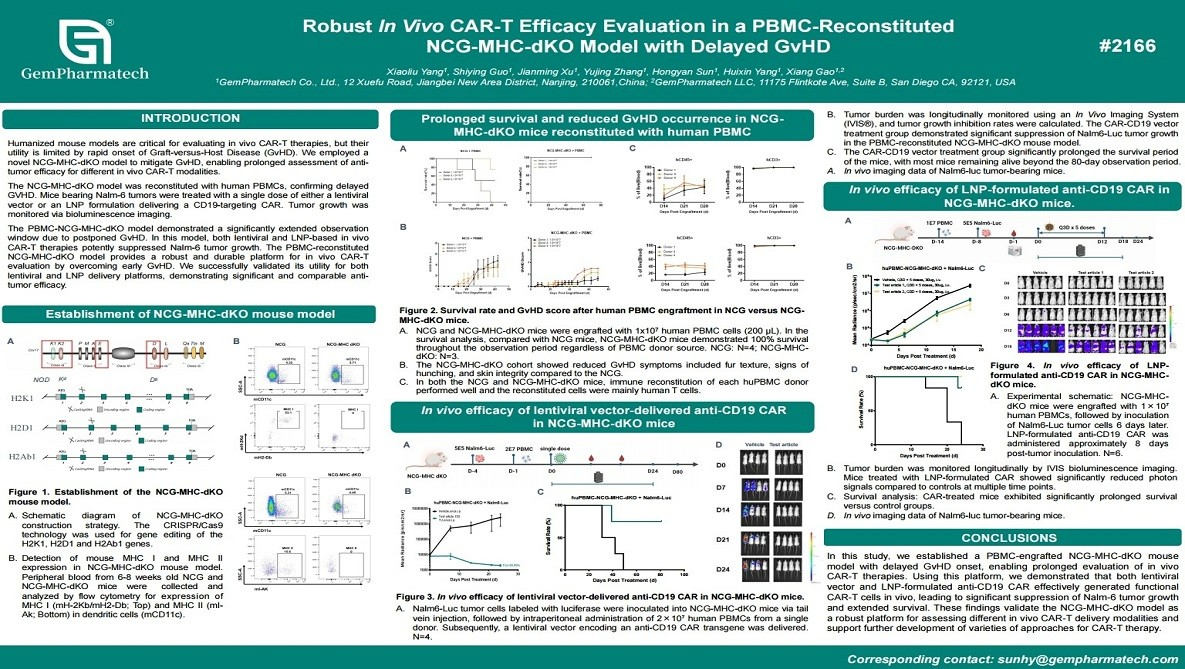
Robust In Vivo CAR-T Efficacy Evaluation in a PBMC-Reconstituted NCG-MHC-dKO Model with Delayed GvHD
Humanized mouse models are widely used for evaluating CAR-T therapies, yet rapid onset of graft-versus-host disease (GvHD) severely restricts long-term studies. GemPharmatech developed a PBMC-reconstituted NCG-MHC-dKO mouse model with significantly delayed GvHD, extending the therapeutic window. In this model, Nalm-6 tumors were engrafted and treated with in vivo CAR-T generation using either lentiviral vectors or lipid nanoparticle (LNP)-delivered CD19-targeting constructs. Tumor burden was monitored by bioluminescence imaging. Both approaches successfully generated functional CAR-T cells in vivo, resulting potent tumor growth inhibition and prolonged survival. This improved model provides a robust and durable platform for comparative assessment of CAR-T therapies.
Download
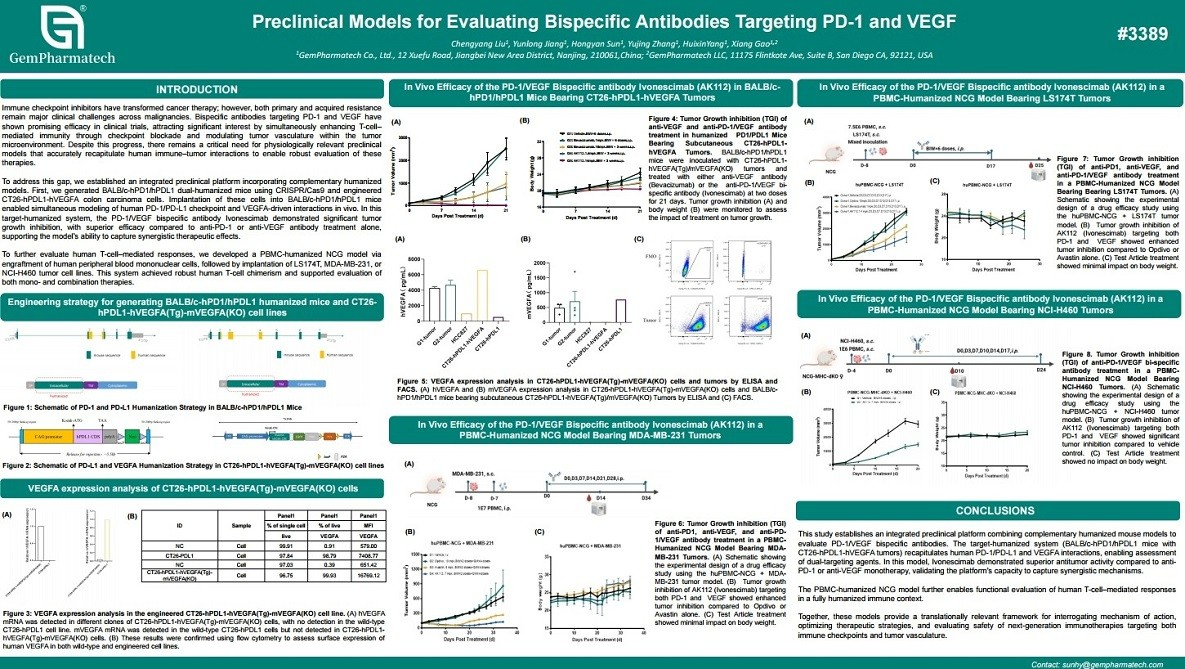
Preclinical Models for Evaluating Bispecific Antibodies Targeting PD-1 and VEGF
Although PD-1 checkpoint inhibitors have transformed cancer treatment, therapeutic resistance remains a significant challenge. Bispecific antibodies co-targeting PD-1 and VEGF offer the potential to simultaneously restore T-cell immunity and inhibit tumor angiogenesis. GemPharmatech developed an integrated preclinical platform using complementary humanized mouse models to evaluate such dual-targeting agents. In a BALB/c-hPD-1/hPD-L1 dual-humanized model bearing CT26-hPD-L1-hVEGFA tumors, the PD-1/VEGF bispecific antibody Ivonescimab demonstrated superior tumor growth inhibition compared to single-pathway blockade. Complementary studies in PBMC-humanized NCG mice further confirmed robust T-cell-mediated antitumor activity across multiple tumor types. This platform enables comprehensive translational evaluation of next-generation PD-1/VEGF bispecific antibodies.
Download
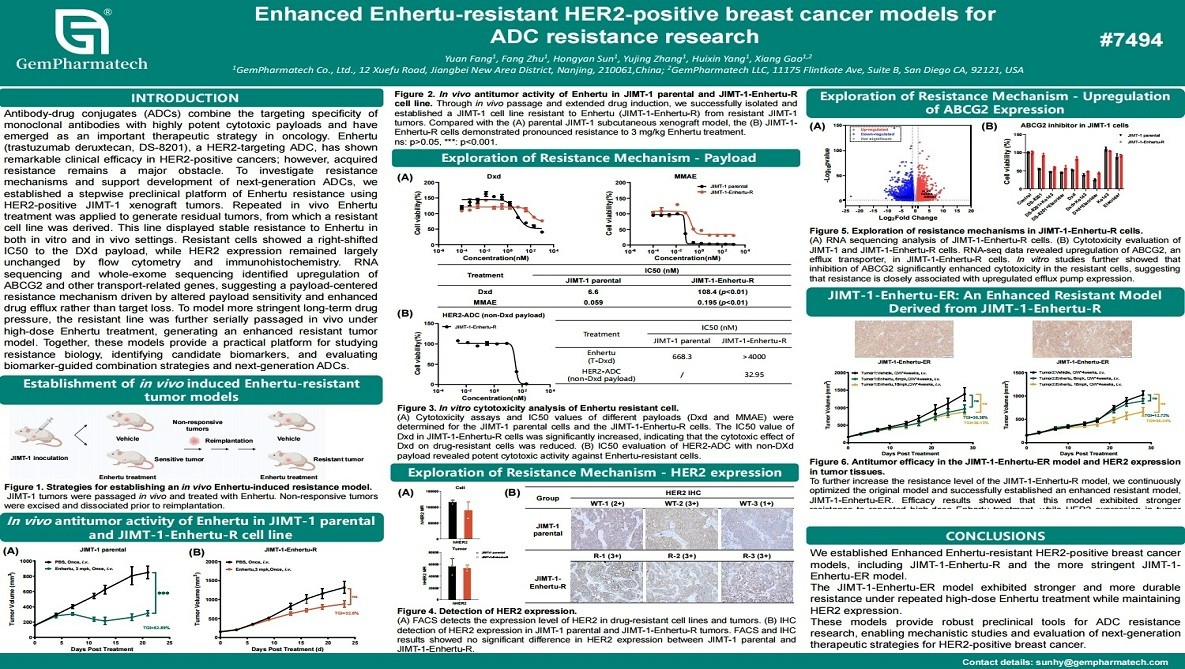
Enhanced Enhertu-Resistant HER2-Positive Breast Cancer Models for ADC Resistance Research
Acquired resistance to HER2-targeted antibody-drug conjugates (ADCs), such as trastuzumab deruxtecan (Enhertu), significantly limits durable clinical benefit in HER2-positive breast cancer. GemPharmatech has established robust Enhertu-resistant models using the HER2-positive JIMT-1 xenograft system through repeated drug exposure. This includes a resistant cell line (JIMT-1-Enhertu-R) and a more stringent in vivo-selected model (JIMT-1-Enhertu-ER). Both models retained high HER2 expression while exhibiting marked resistance to the DXd payload, associated with upregulation of drug efflux transporters including ABCG2. These well-characterized models provide valuable platforms for elucidating ADC resistance mechanisms and evaluating next-generation HER2-targeted therapies.
Download
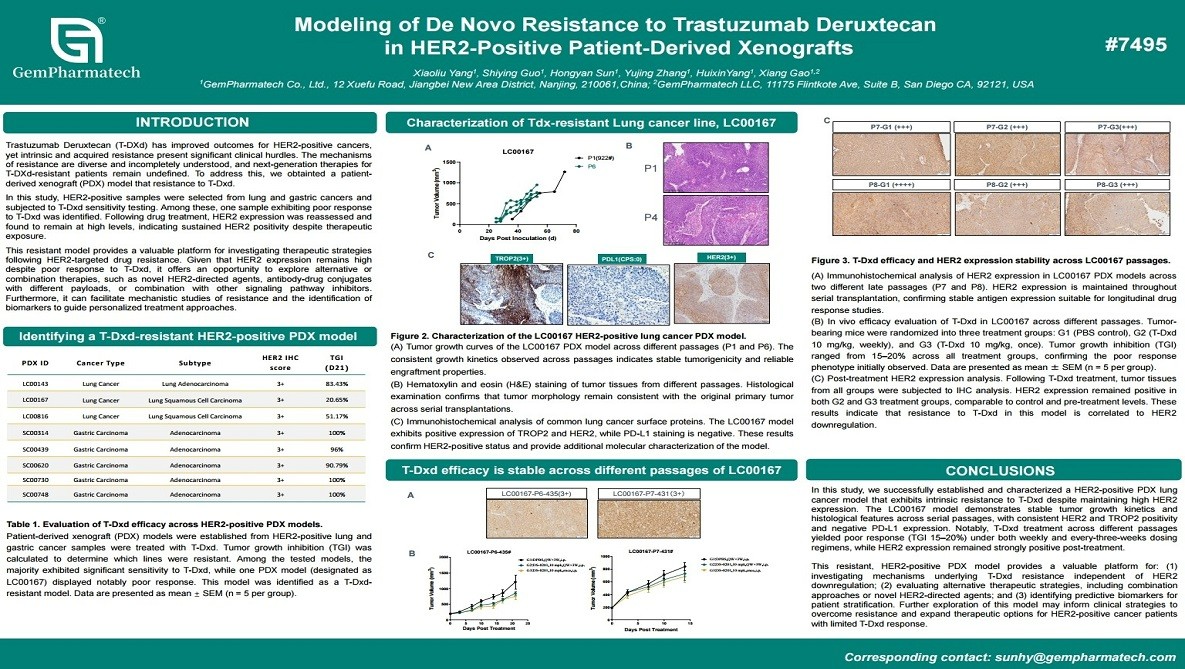
Modeling of De Novo Resistance to Trastuzumab Deruxtecan in HER2-Positive Patient-Derived Xenografts
Resistance to trastuzumab deruxtecan (T-DXd) in HER2-positive cancers represents a significant unmet clinical need. GemPharmatech has established a patient-derived xenograft (PDX) model of intrinsic T-DXd resistance derived from lung and gastric cancers. This model maintains high HER2 expression yet demonstrates robust resistance to T-DXd in vivo. The persistence of HER2 without target loss strongly suggests alternative resistance mechanisms independent of antigen downregulation. This clinically relevant PDX model provides a valuable platform for dissecting T-DXd resistance biology, identifying predictive biomarkers, and evaluating novel therapeutic strategies or combinations for HER2-positive tumors.
Download
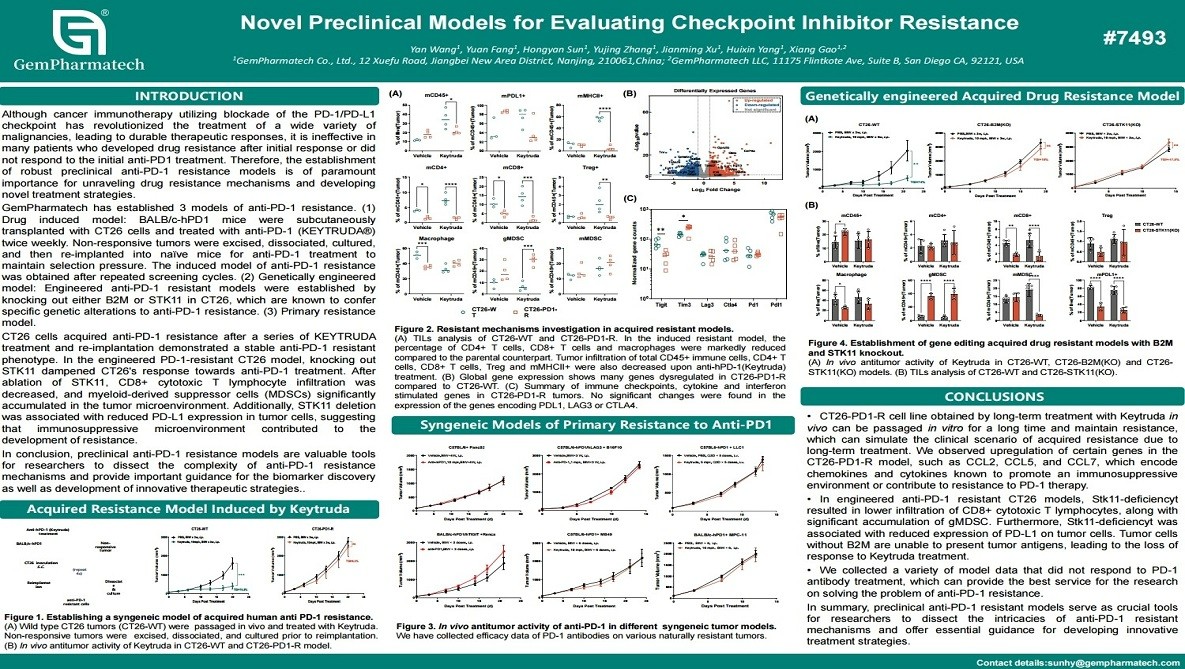
Novel Preclinical Models for Evaluating Checkpoint Inhibitor Resistance
Resistance to PD-1/PD-L1 checkpoint inhibitors remains a major obstacle in cancer immunotherapy. GemPharmatech has developed a comprehensive panel of preclinical anti-PD-1 resistance models, including drug-induced, genetically engineered, and primary resistance systems. Drug-induced resistance was established through chronic anti-PD-1 treatment and serial tumor re-implantation in BALB/c-hPD-1 humanized mice. Genetically engineered models included CT26 tumors with B2M or STK11 knockout. These resistant tumors exhibited distinct immunosuppressive microenvironments, characterized by reduced CD8⁺ T-cell infiltration and expansion of suppressive myeloid cells. Collectively, these well-characterized models serve as valuable translational tools to dissect resistance mechanisms, identify biomarkers, and develop next-generation immunotherapy strategies.
Download
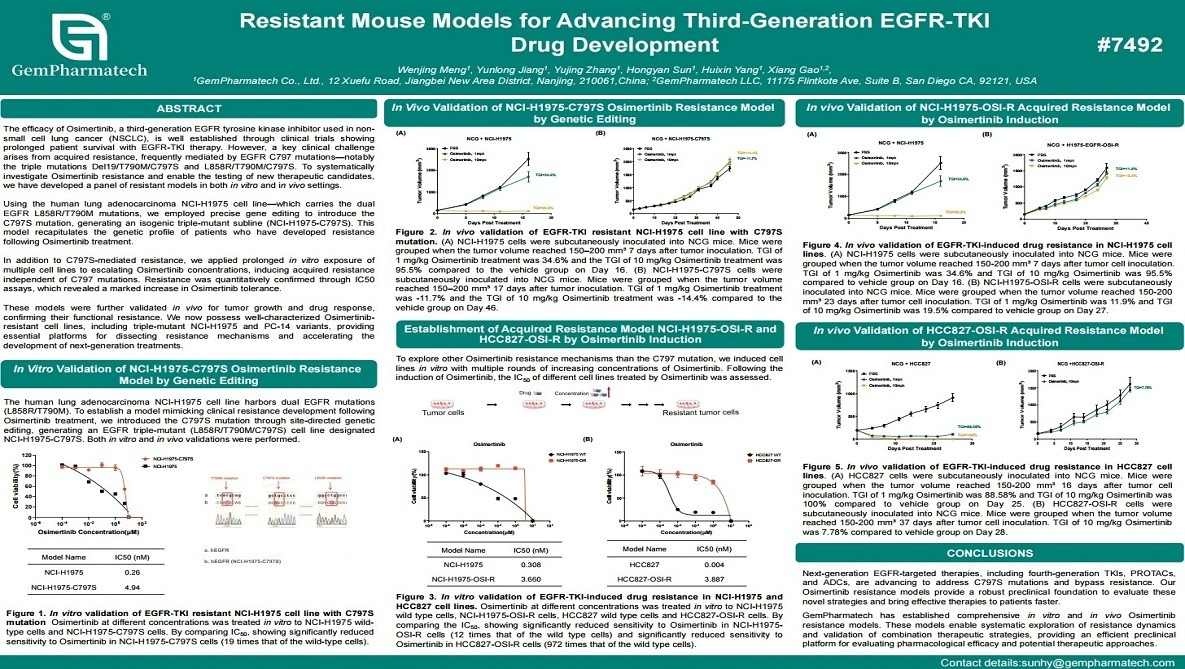
Resistant Mouse Models for Advancing Third-Generation EGFR-TKI Drug Development
Osimertinib, a third-generation EGFR tyrosine kinase inhibitor (TKI), has transformed the treatment of EGFR-mutant non-small cell lung cancer (NSCLC). However, acquired resistance mediated by EGFR C797 mutations remains a major unmet clinical need. GemPharmatech has developed a panel of Osimertinib-resistant preclinical models using both gene-editing and chronic drug-exposure strategies. This includes the NCI-H1975-C797S triple-mutant cell line (EGFR L858R/T790M/C797S), along with additional resistant lines generated through prolonged osimertinib exposure. These models were thoroughly validated in vitro and in vivo, exhibiting stable and robust resistance phenotypes. Together, they provide powerful translation platforms for elucidating resistance mechanisms and evaluating next-generation EGFR-targeted therapies.
Download
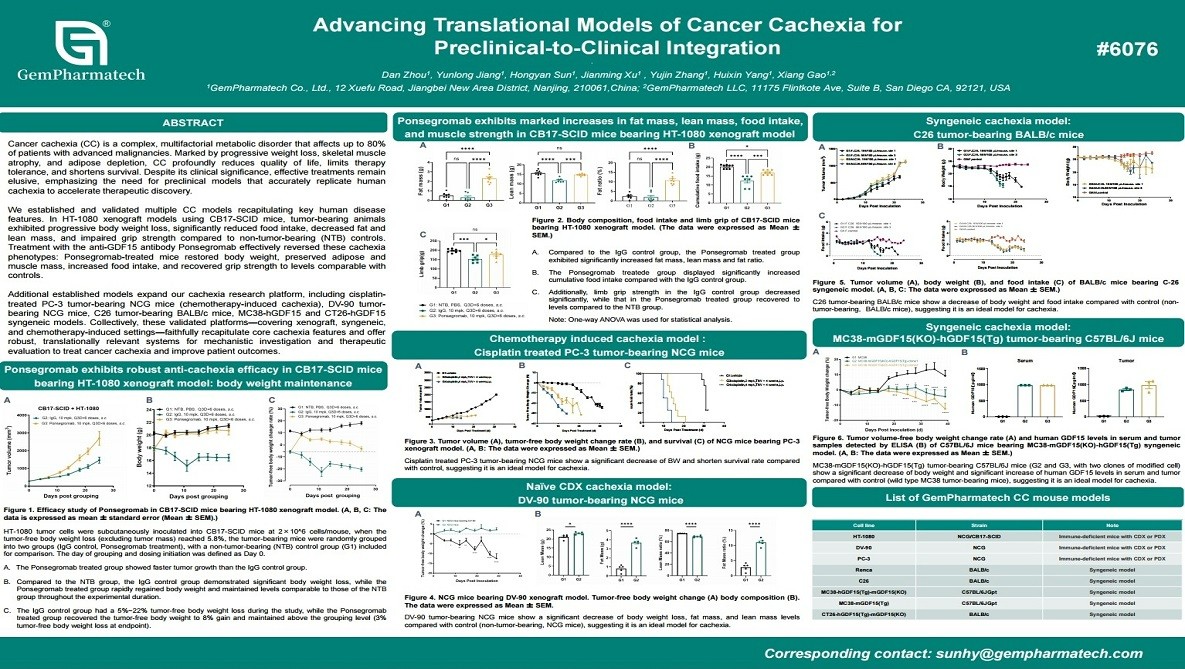
Advancing Translational Models of Cancer Cachexia for Preclinical-to-Clinical Integration
Cancer cachexia is a devastating metabolic syndrome characterized by progressive weight loss, skeletal muscle wasting, and reduced tolerance to anticancer therapies. GemPharmatech has established and validated multiple preclinical models that closely recapitulate key features of human cancer cachexia. In HT-1080 xenograft models, tumor-bearing mice exhibited progressive body weight loss, anorexia, and depletion of muscle and fat mass. Treatment with the anti-GDF15 neutralizing antibody Ponsegromab effectively reversed these cachectic symptoms. Complementary syngeneic, xenograft, and chemotherapy-induced cachexia models further confirmed the robustness of these platforms. These well-characterized models provide valuable translational tools for elucidating cachexia mechanisms and evaluating novel therapeutic interventions.
Download
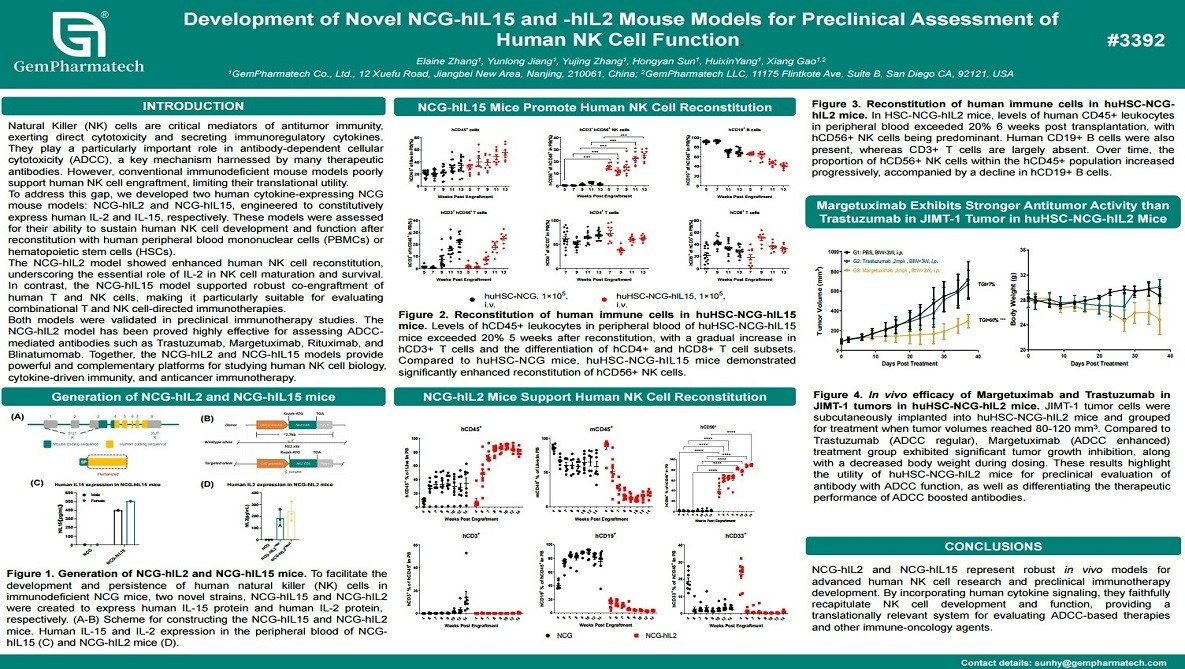
Development of Novel NCG-hIL15 and -hIL2 Mouse Models for Preclinical Assessment of Human NK Cell Function
Natural killer (NK) cells are critical effectors of innate antitumor immunity, yet conventional immunodeficient mouse models poorly support human NK cell engraftment and function. To overcome this limitation, GemPharmatech developed NCG-hIL2 and NCG-hIL15 humanized mouse models expressing human IL-2 or IL-15, respectively. Following reconstitution with human PBMCs or hematopoietic stem cells, both models markedly enhanced human NK cell development. Notably, the NCG-hIL15 model further enabled robust co-engraftment of T cells and NK cells. These models effectively supported the evaluation of NK cell-mediated immunotherapies, including antibody-dependent cellular cytotoxicity (ADCC)-inducing antibodies. Together, they provide valuable complementary platforms for studying human NK cell biology and advancing NK-based cancer immunotherapies.
Download
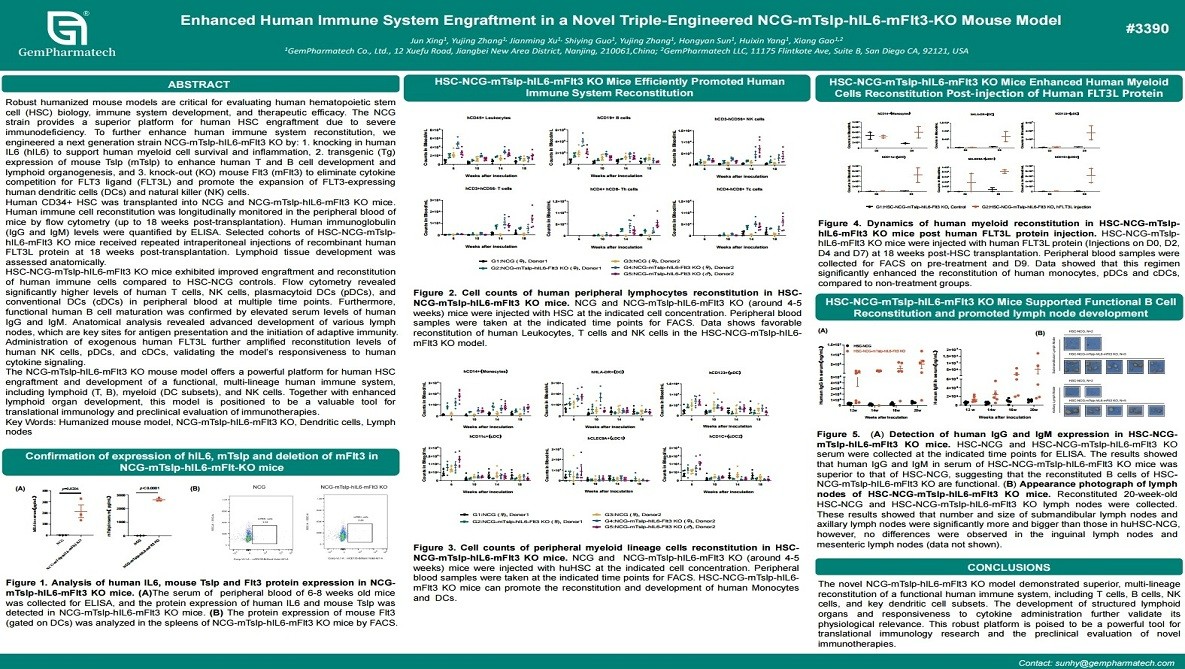
Enhanced Human Immune System Engraftment in a Novel Triple-Engineered NCG-mTslp-hIL6-mFlt3-KO Mouse Model
Robust humanized mouse models are critical for studying human hematopoietic stem cell (HSC) engraftment and immune system reconstitution. GemPharmatech developed a novel triple-engineered NCG-mTslp-hIL6-mFlt3 KO mouse model by introducing human IL-6, expressing mouse TSLP, and knocking out mouse Flt3 to optimize human immune development. Following human CD34+ HSC transplantation, the model exhibited significantly enhanced reconstitution of T cells, B cells, NK cells, and dendritic cell subsets, accompanied by improved lymphoid tissue development. Administration of human FLT3L further boosted immune cell expansion. This advanced model provides a superior platform for immunology research and preclinical evaluation of immune-based therapies.
Download
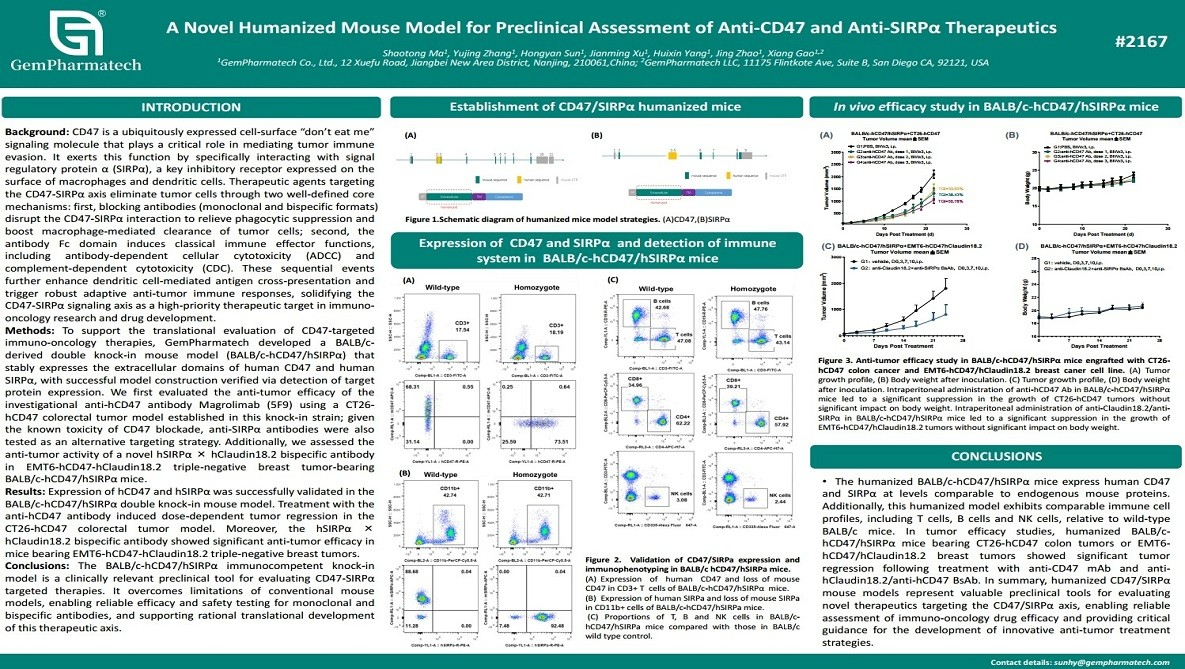
A Novel Humanized Mouse Model for Preclinical Assessment of Anti-CD47 and Anti-SIRPα Therapeutics
CD47–SIRPα signaling is a critical myeloid checkpoint that enables tumor cells to evade macrophage phagocytosis, representing a promising target for cancer immunotherapy. To enable preclinical evaluation of this pathway, GemPharmatech developed a BALB/c-based double humanized knock-in mouse model (BALB/c-hCD47/hSIRPα) expressing the extracellular domains of human CD47 and SIRPα. Using CT26-hCD47 colorectal and EMT6-hCD47-hClaudin18.2 triple-negative breast cancer models, the anti-CD47 antibody Magrolimab (5F9) induced dose-dependent tumor regression. Additionally, a bispecific anti-SIRPα × anti-Claudin18.2 antibody demonstrated potent anti-tumor efficacy. This fully immunocompetent model provides a valuable platform for evaluating both monoclonal and bispecific antibodies targeting the CD47–SIRPα axis.
Download
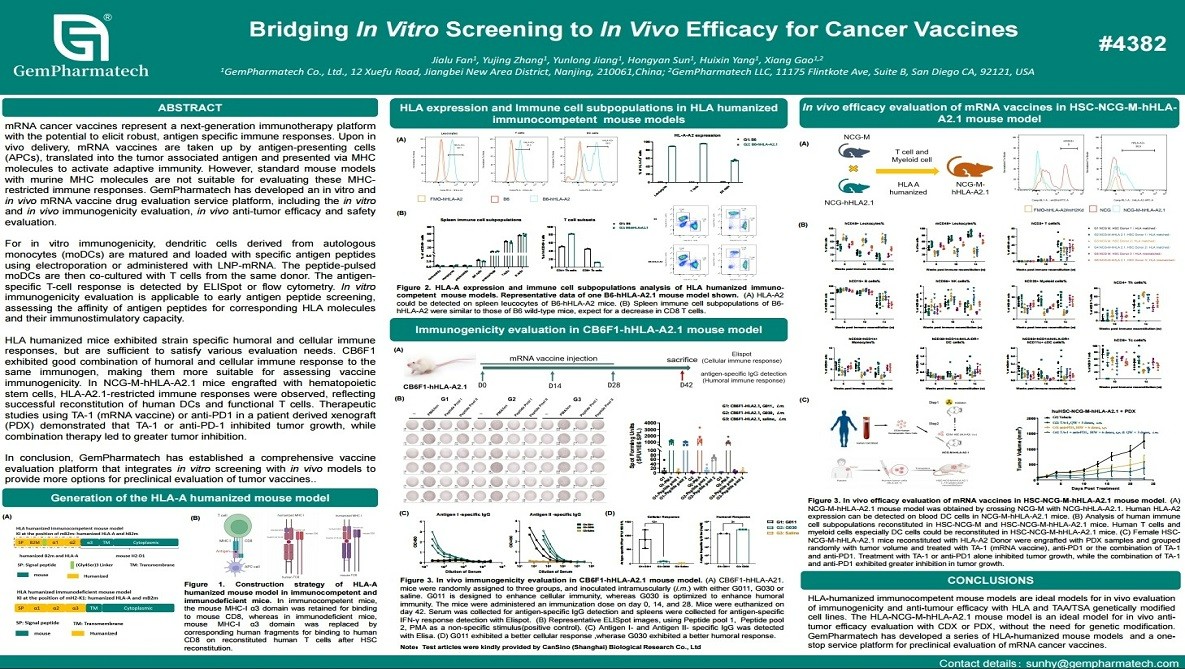
Bridging In Vitro Screening to In Vivo Efficacy for Cancer Vaccines
mRNA cancer vaccines induce antigen-specific immunity by promoting dendritic cell-mediated antigen presentation and T-cell activation, yet their preclinical evaluation requires models that faithfully recapitulate human MHC-restricted immune responses. GemPharmatech developed an integrated platform for comprehensive assessment of mRNA cancer vaccines, combining in vitro immunogenicity assays with in vivo efficacy and safety studies. In vitro, monocyte-derived dendritic cells co-cultured with autologous T cells were used to evaluate antigen-specific immune activation. In vivo, HLA-humanized mouse models, including HLA-A2.1 mice, enabled the assessment of human-relevant T-cell responses. Therapeutic studies demonstrated potent tumor growth inhibition by mRNA vaccines, with further enhancement when combined with anti-PD-1 therapy in xenograft models. This platform provides a robust translational tool for optimizing mRNA-based cancer vaccines.
Download
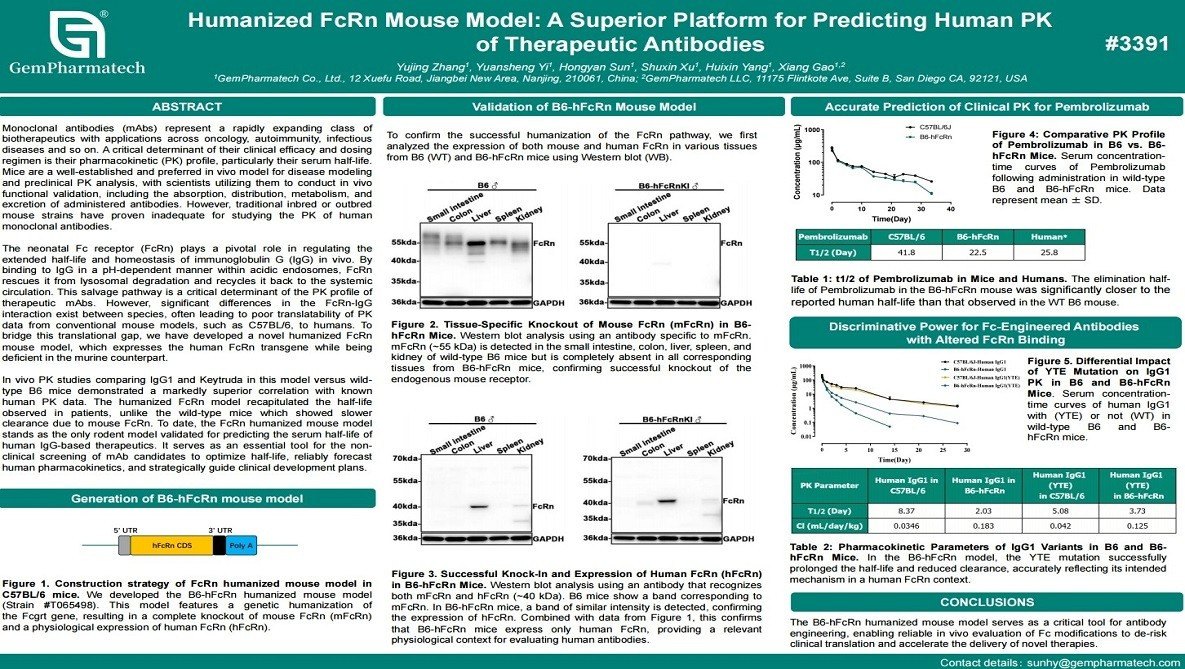
Humanized FcRn Mouse Model: A Superior Platform for Predicting Human PK of Therapeutic Antibodies
Accurate prediction of pharmacokinetics (PK) is critical for monoclonal antibody development, yet conventional mouse models often fail to recapitulate human IgG half-life due to species differences in FcRn binding. GemPharmatech has developed a humanized FcRn mouse model expressing the human FcRn transgene on a murine FcRn-deficient background. In vivo PK studies with human IgG1 and pembrolizumab (Keytruda) demonstrated significantly improved correlation with clinical human data, including more accurate serum half-life compared to wild-type mice. This humanized FcRn model offers a robust and translational platform for preclinical PK evaluation and optimization of therapeutic antibodies.
Download
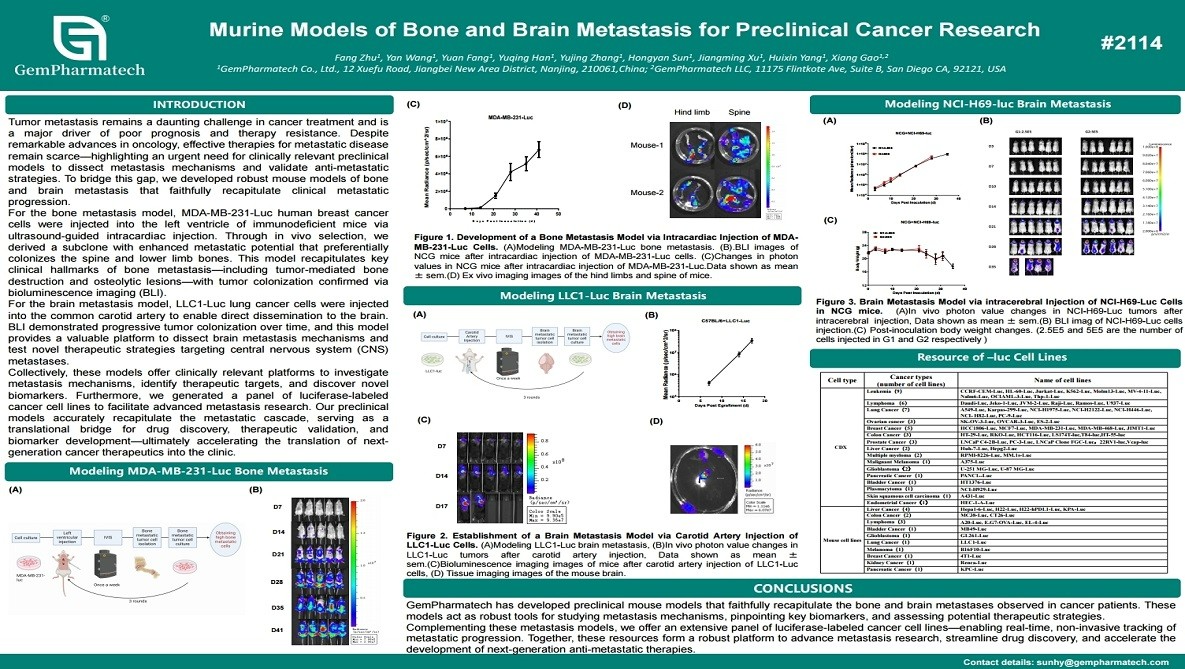
Murine Models of Bone and Brain Metastasis for Preclinical Cancer Research
Tumor metastasis is the primary driver of cancer mortality and therapy resistance, highlighting urgent need for clinically relevant preclinical models. GemPharmatech has developed robust murine models for bone and brain metastasis. Bone metastasis was established via intracardiac injection of MDA-MB-231-Luc cells, followed by in vivo selection of a highly bone-tropic subclone, confirmed by bioluminescence imaging and osteolytic lesions. Brain metastasis was induced through carotid artery injection of LLC1-Luc cells, enabling efficient tumor dissemination to the brain. These models provide platforms for studying metastatic mechanisms and evaluating novel therapies, advancing translational cancer research.
Download
Previous:N/A